
VAERS data released Friday by the Centers for Disease Control and Prevention included a total of 1,071,856 reports of adverse events from all age groups following COVID vaccines, including 22,607 deaths and 178,994 serious injuries between Dec. 14, 2020, and Jan. 21, 2022.
The Centers for Disease Control and Prevention (CDC) today released new data showing a total of 1,071,856 reports of adverse events following COVID vaccines were submitted between Dec. 14, 2020, and Jan. 21, 2022, to the Vaccine Adverse Event Reporting System (VAERS). VAERS is the primary government-funded system for reporting adverse vaccine reactions in the U.S.
The data included a total of 22,607 reports of deaths — an increase of 414 over the previous week — and 178,994 reports of serious injuries, including deaths, during the same time period — up 4,130 compared with the previous week.
Excluding “foreign reports” to VAERS, 740,000 adverse events, including 10,316 deaths and 67,496 serious injuries, were reported in the U.S. between Dec. 14, 2020, and Jan. 21, 2022.
Foreign reports are reports foreign subsidiaries send to U.S. vaccine manufacturers. Under U.S. Food and Drug Administration (FDA) regulations, if a manufacturer is notified of a foreign case report that describes an event that is both serious and does not appear on the product’s labeling, the manufacturer is required to submit the report to VAERS.
Of the 10,316 U.S. deaths reported as of Jan. 21, 19% occurred within 24 hours of vaccination, 24% occurred within 48 hours of vaccination and 61% occurred in people who experienced an onset of symptoms within 48 hours of being vaccinated.
In the U.S., 532.4 million COVID vaccine doses had been administered as of Jan. 21, including 312 million doses of Pfizer, 202 million doses of Moderna and 19 million doses of Johnson & Johnson (J&J).
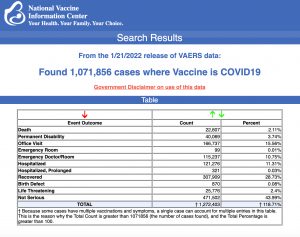
Every Friday, VAERS publishes vaccine injury reports received as of a specified date. Reports submitted to VAERS require further investigation before a causal relationship can be confirmed. Historically, VAERS has been shown to report only 1% of actual vaccine adverse events.
U.S. VAERS data from Dec. 14, 2020, to Jan. 21, 2022, for 5- to 11-year-olds show:
- 7,052 adverse events, including 152 rated as serious and 3 reported deaths.
The most recent death involves a 7-year-old girl (VAERS I.D. 1975356) from Minnesota who died 11 days after receiving her first dose of Pfizer’s COVID vaccine when she was found unresponsive by her mother. An autopsy is pending.
- 14 reports of myocarditis and pericarditis (heart inflammation).
- 24 reports of blood clotting disorders.
U.S. VAERS data from Dec. 14, 2020, to Jan. 21, 2022, for 12- to 17-year-olds show:
- 27,772 adverse events, including 1,588 rated as serious and 37 reported deaths.
The most recent deaths involve a 13-year-old male (VAERS I.D. 2042005) from an unidentified state who died from a sudden heart attack seven months after receiving his second dose of Moderna, and a 17-year-old female from an unidentified state (VAERS I.D. 2039111) who died after receiving her first dose of Moderna. Medical information was limited and it is unknown if an autopsy was performed in either case.
- 68 reports of anaphylaxis among 12- to 17-year-olds where the reaction was life-threatening, required treatment or resulted in death — with 96% of cases attributed to Pfizer’s vaccine.
- 609 reports of myocarditis and pericarditis with 597 cases attributed to Pfizer’s vaccine.
- 154 reports of blood clotting disorders, with all cases attributed to Pfizer.
U.S. VAERS data from Dec. 14, 2020, to Jan. 21, 2022, for all age groups combined, show:
- 21% of deaths were related to cardiac disorders.
- 54% of those who died were male, 41% were female and the remaining death reports did not include the gender of the deceased.
- The average age of death was 72.7.
- As of Jan. 21, 4,925 pregnant women reported adverse events related to COVID vaccines, including 1,575 reports of miscarriage or premature birth.
- Of the 3,474 cases of Bell’s Palsy reported, 51% were attributed to Pfizer vaccinations, 41% to Moderna and 8% to J&J.
- 850 reports of Guillain-Barré syndrome (GBS), with 41% of cases attributed to Pfizer, 30% to Moderna and 28% to J&J.
- 2,281 reports of anaphylaxis where the reaction was life-threatening, required treatment or resulted in death.
- 12,704 reports of blood clotting disorders in the U.S. Of those, 5,646 reports were attributed to Pfizer, 4,521 reports to Moderna and 2,490 reports to J&J.
- 1,542 reports of myocardial infarction.
- 3,817 cases of myocarditis and pericarditis with 2,348 cases attributed to Pfizer, 1,293 cases to Moderna and 164 cases to J&J’s COVID vaccine.
Unvaccinated man denied heart transplant by Boston hospital
DJ Ferguson, 31, was removed from the top of a heart transplant at Boston’s Brigham and Women’s Hospital because he was not vaccinated against COVID.
Ferguson on Tuesday received a mechanical heart pump — called a left ventricular assist device — that should keep him alive for up to five years, but he won’t have much of a life, his father said.
According to ABC News, Ferguson, a father of two children with another baby on the way, didn’t want the vaccine because he feared it would complicate his heart condition. He also said getting vaccinated would go against his basic principles.
“The organs are scarce, we are not going to distribute them to someone who has a poor chance of living when others who are vaccinated have a better chance post-surgery of surviving,” Dr. Arthur Caplan, who runs Medical Ethics at NYU Grossman School of Medicine told MassLive.
Despite the open-heart surgery, Ferguson still needs a transplant due to his rapid deterioration, Ferguson’s parents told “Tucker Carlson Tonight” on Wednesday.
COVID vaccine regime for children under age 4 will include 3 doses, Fauci says
White House chief medical advisor Dr. Anthony Fauci on Wednesday said the COVID vaccine regime for kids younger than 4 years old will likely include three doses when it’s authorized because two shots did not induce an adequate immune response in 2- to 4-year-olds in Pfizer’s clinical trials.
“Dose and regimen for children 6 months to 24 months worked well, but it turned out the other group from 24 months to 4 years did not yet reach the level of non-inferiority, so the studies are continued,” Fauci said, referencing effectiveness standard comparison to adults.
Fauci said he hopes the U.S. Food and Drug Administration will authorize the Pfizer and BioNTech COVID vaccine for children under 5 years old next month, although he can’t say for sure when the agency will render its decision.
Sweden decides against COVID vaccines for children 5 to 11
Sweden won’t recommend COVID vaccines for kids under 12 years old because the benefits did not outweigh the risks, but will “constantly” reassess the situation, Reuters reported.
The Public Health Agency of Sweden said in a press release on Thursday the medical benefit for a child aged 5-11 who has received a vaccine against COVID “is currently small.”
Britta Bjorkholm, a Sweden health official, said during a news conference, “With the knowledge we have today, with a low risk for serious disease for kids, we don’t see any clear benefit with vaccinating them.”
Karin Tegmark Wisell, director-general of the Public Health Agency of Sweden, said updated guidance would be provided prior to the fall term.
COVID vaccines causing miscarriages, cancer, neurological disorders among Military
In a hearing organized this week by Sen. Ron Johnson (R-Wis.), attorney Thomas Renz told a panel of experts data provided to him by three whistleblowers show COVID vaccines are causing catastrophic harm to members of the U.S. military while not preventing them from getting the virus.
Renz summarized data obtained from the Defense Medical Epidemiology Database — the military’s longstanding epidemiological database of service members.
The data show miscarriages and cancer increased 300% in 2021 over the previous five-year average. Neurological disorders increased 1000% in 2021 over the past five-year average, increasing from 82,000 to 863,000 in one year.
“Our soldiers are being experimented on, injured and sometimes possibly killed,” Renz said.
Following Renz’s presentation, attorney Leigh Dundas reported evidence of the DOD doctoring data in DMED to conceal cases of myocarditis in service members vaccinated for COVID.
OSHA withdraws COVID vaccine mandate
The U.S. Department of Labor this week announced it is withdrawing the Biden administration’s COVID vaccine-or-test mandate for large employers.
In pulling the rule, the department said it recognized the Emergency Temporary Standard could not be revived after the U.S. Supreme Court blocked it earlier this month and will plan instead to set a permanent standard for the vaccine mandate, according to a notice provided to the court by the Occupational Safety and Health Administration (OSHA).
The Labor Department’s decision to withdraw the rule means pending legal proceedings in the 6th Circuit will be dropped.
OSHA could move a version of the vaccine-or-test rule through its rule-making process, but would still likely face legal challenges.